JAMA-(ENEWSPF)- A study just released by the Journal of the American Medical Association concluded that the once-touted hydroxychloroquine made no significant difference in patients who were hospitalized with COVID-19. The drug made no difference in outcomes of patients regardless of the patients’ age, sex, race/ethnicity, baseline illness severity, and duration of symptoms.
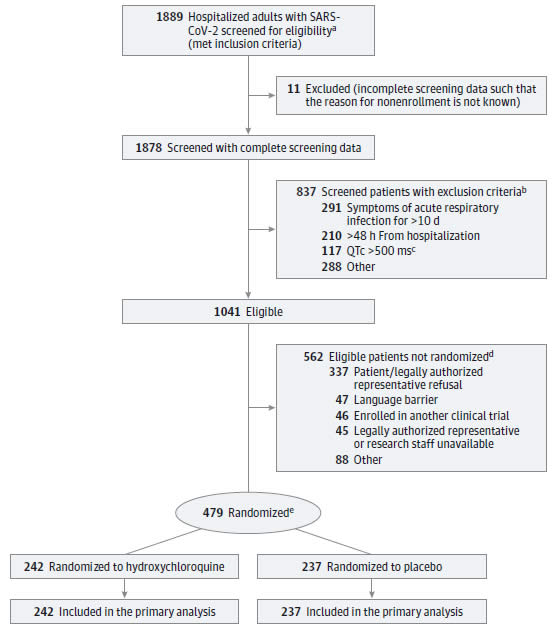
According to the authors of the study, “In this multicenter, blinded, placebo-controlled randomized clinical trial conducted at 34 US hospitals, treatment with hydroxychloroquine did not improve or worsen clinical out- comes for adults hospitalized for respiratory illness from COVID-19. These findings were consistent in all subgroups and for all outcomes evaluated, including an ordinal scale of clinical status, mortality, organ failures, duration of oxygen use, and hospital length of stay.”
“The finding of this clinical trial that hydroxychloroquine was not efficacious for the treatment of COVID-19 is consistent with results from recent in vitro studies suggesting no antiviral activity for hydroxychloroquine against SARS-CoV-2 and open-label pragmatic trials in the United Kingdom and Brazil suggesting no clinical benefit. Interpreted along with these prior studies, the results of this trial provide strong evidence that hydroxychloroquine is not beneficial for adults hospitalized with COVID-19,” the study concludes.
“Strengths of this trial included its blinded, placebo-controlled design, high adherence to the study protocol, rigorous monitoring for safety events and adverse events, and rapid recruitment from geographically diverse hospitals serving ethnically and racially diverse populations within the US. Additionally, the primary outcome was a patient-centered, clinically meaningful ordinal scale that captured mortality and morbidity related to COVID-19,” according to the study’s authors.